BRN 0085894
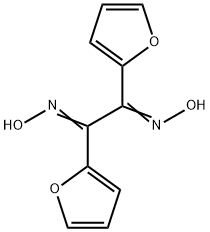
di-2-furylethanedione dioxime
CAS: 522-27-0
Molecular Formula: C10H8N2O4
BRN 0085894 - Names and Identifiers
BRN 0085894 - Physico-chemical Properties
| Molecular Formula | C10H8N2O4 |
| Molar Mass | 220.18 |
| Density | 1.3848 (rough estimate) |
| Melting Point | 155 °C |
| Boling Point | 361.11°C (rough estimate) |
| Flash Point | 159.7°C |
| Vapor Presure | 3.32E-05mmHg at 25°C |
| Merck | 14,4306 |
| BRN | 85894 |
| pKa | 9.00±0.10(Predicted) |
| Storage Condition | −20°C |
| Refractive Index | 1.5910 (estimate) |
| Physical and Chemical Properties | White needle crystal. Melting point 166-168 ℃ (anhydrous),84-85 ℃ (containing crystal water). Easily soluble in alcohol and ether, slightly soluble in benzene and petroleum ether, hardly soluble in water. |
BRN 0085894 - Risk and Safety
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| RTECS | LV0590000 |
| TSCA | Yes |
BRN 0085894 - Reference Information
| chemical properties | white needle crystal. Melting point 166-168 ℃ (anhydrous),84-85 ℃ (containing crystal water). Easily soluble in alcohol and ether, slightly soluble in benzene and petroleum ether, hardly soluble in water. |
| Uses | This product can form stable complexes with nickel, platinum, palladium, rhenium, etc., and is used as a chelating extractant to extract and separate nickel, palladium, etc. |
| production method | is prepared from furanoyl (see previous article). Mix water with 8 times the weight of the group and excess hydroxylamine hydrochloride and heat it on a water bath until it is completely dissolved. Pour the solution into boiling water 6 times the base volume and keep boiling for a few minutes. Cooling, filtering out the precipitate, washing with a large amount of water, and then evaporating and drying at 30-90 ℃ is the finished product. |
| category | toxic substances |
| toxicity classification | poisoning |
| acute toxicity | vein-mouse LDL0: 180 mg/kg |
| flammability hazard characteristics | flammability; thermal decomposition of toxic nitrogen oxide smoke |
| storage and transportation characteristics | warehouse ventilation and low temperature drying |
| fire extinguishing agent | dry powder, foam, sand, carbon dioxide, mist water |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
Last Update:2024-04-09 21:04:16
Supplier List
Product Name: ALPHA-FURIL DIOXIME Request for quotation
CAS: 522-27-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 522-27-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: ALPHA-FURIL DIOXIME Request for quotation
CAS: 522-27-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 522-27-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
View History
